 An aprotic solvent is one that does not form hydrogen bonds. A protic solvent is one that has a hydrogen atom bound to an oxygen or nitrogen. Polar solvents can be divided into protic or aprotic. Protic solvents are favored for this reaction: Acetic Acid, tert-Butyl alcohol, Ethanol, Methanol, Formic Acid and Water. Sn1 reactions - the substrate dissociates into a carbocation ( formed as an intermediate) in a slow reaction followed by a fast attack by the nucleophile. Rates of reaction: Alkyl halide > primary > secondary Aprotic solvents are favored for this reaction: Hexane, Benzene, Diethyl Ether, Chloroform, Ethyl Acetate, Acetone, HMPA, DMF, DMSO. Favored for primary and secondary substrates. Look for these compounds as nucleophiles: Hydride, Cyanide, Iodide, Hydroxide, Methoxide, Chloride, Acetate, Ammonia. Sn2 reactions - nucleophile and alkyl halide reaction occurs with complete inversion of steriochemistry at the chiral carbon center. 
An Electrophile (usually an acid, alkyl halides ) is electron poor and will accept electrons from the nucleophile. 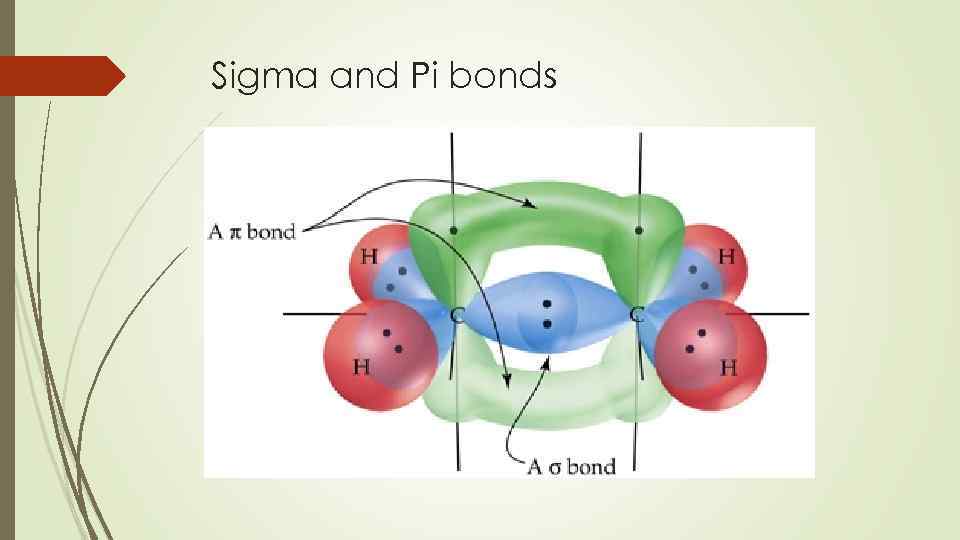
The electron pair movement is designated with curved arrows, which shows the motion of the electrons.Ī Nucleophile (usually a base, carbon=carbon ) is a reagent that is electron rich and can form a bond by donating a pair of electrons to an electron poor site. Heterolytic - polar compound is formed, where 2 electrons stay with one fragmentĬarbon has a partial positive charge when bonded to O,N,F,Cl or Br.īonds are made when an electron rich reagent donates a pair of electrons to the electron deficient reagent.īonds are broken when either pair leaves with the electron pair. Homolytic - free radical is formed, where one electron stays with each fragment Two ways in which a covalent bond can break: 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
